The Development of the Periodic Table
The entire science of chemistry -- although at this time, it was just a "patchwork of observations and discoveries" -- was centered around the elements. Mendeleev noticed that there were certain patterns between different groups of elements. He wondered if anything would be revealed if he could somehow organize them. He wrote down the names of the 65 known elements on cards (one element on each) and wrote the chemical and physical properties of each element, including atomic weight. He moved the cards around for hours until he fell asleep at his desk.
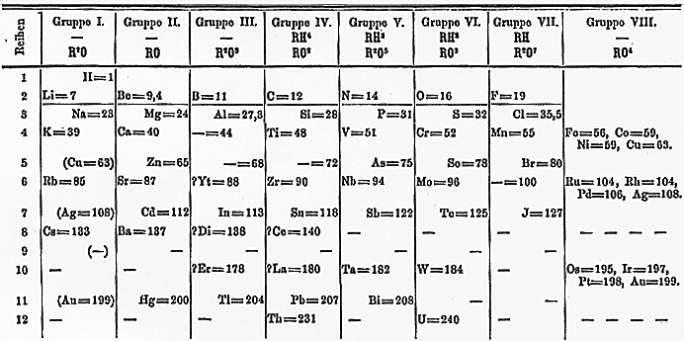
When he awoke, he realized what the pattern was. By arranging the elements according to their atomic mass, he could organize them into groups of elements that possessed similar properties. He wrote down the arrangement right away. After that, it took him two weeks to publish The Relation between the Properties and the Atomic Weights of the Elements. Mendeleev's periodic table had finally been introduced to the world.
Many people had created periodic tables before Mendeleev. For example, Lothar Meyer created a rough periodic table in 1864. In 1868, he devised one that was similiar to Mendeleev's. However, he didn't publish his until 1870, the year after Mendeleev published his own periodic table.
John Newlands published his own periodic table in 1865. He even wrote his own law: "Any given element will exhibit analogous behavior to the eighth element following it in the table." Unfortunately, Newlands' work was ignored.
But why was only Mendeleev's table successful? His table proposed that some elements had their atomic weights measured incorrectly, since their behaviors did not agree with his prediction. In addition, he predicted the existence of eight new elements and their properties, even before they actually existed.
Mendeleev's periodic table has paved the way for modern chemistry. If it weren't for Mendeleev's observations and creation, it is likely that we wouldn't have a Periodic Table today. Because he noticed these recurring patterns in the behavior of elements, chemists today have been able to continuously discover and add more elements to the Periodic Table.
When he awoke, he realized what the pattern was. By arranging the elements according to their atomic mass, he could organize them into groups of elements that possessed similar properties. He wrote down the arrangement right away. After that, it took him two weeks to publish The Relation between the Properties and the Atomic Weights of the Elements. Mendeleev's periodic table had finally been introduced to the world.
Many people had created periodic tables before Mendeleev. For example, Lothar Meyer created a rough periodic table in 1864. In 1868, he devised one that was similiar to Mendeleev's. However, he didn't publish his until 1870, the year after Mendeleev published his own periodic table.
John Newlands published his own periodic table in 1865. He even wrote his own law: "Any given element will exhibit analogous behavior to the eighth element following it in the table." Unfortunately, Newlands' work was ignored.
But why was only Mendeleev's table successful? His table proposed that some elements had their atomic weights measured incorrectly, since their behaviors did not agree with his prediction. In addition, he predicted the existence of eight new elements and their properties, even before they actually existed.
Mendeleev's periodic table has paved the way for modern chemistry. If it weren't for Mendeleev's observations and creation, it is likely that we wouldn't have a Periodic Table today. Because he noticed these recurring patterns in the behavior of elements, chemists today have been able to continuously discover and add more elements to the Periodic Table.